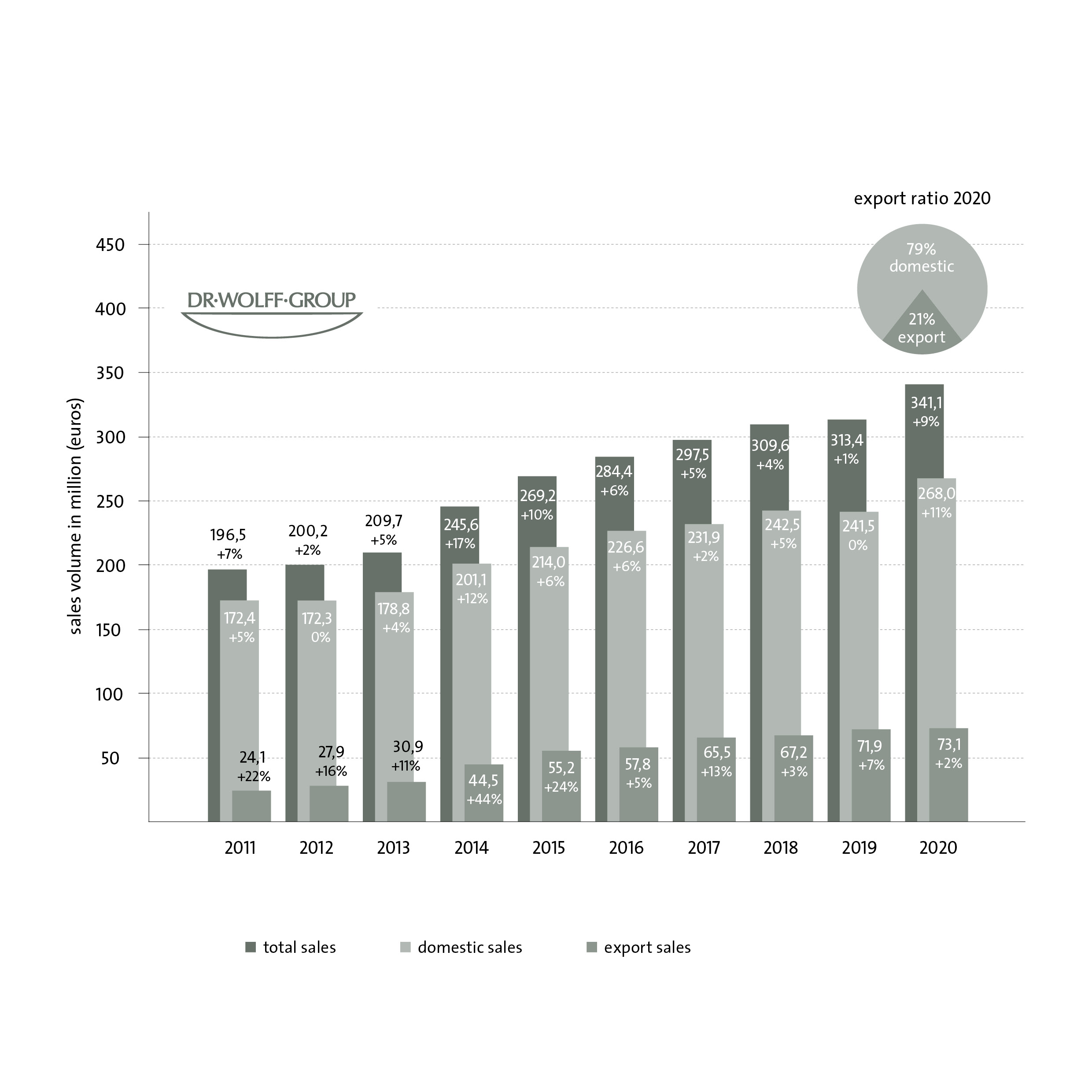
In 2020, Bielefeld-based Dr. Wolff Group recorded a turnover of 341.1 million euros (2019: 313.4 million euros), an increase of about nine percent, with exports continuing to account for more than 20 percent. Decades of evidence-based research with numerous scientific publications ensure highest product quality. Thanks to the trust of millions of consumers coupled with bold and fast action, this course could be continued in the first quarter of 2021 with more than three percent growth.
“Crisis in the morning, opportunity in the afternoon”.
In 2020, the family business had to act flexibly and quickly. The focus was on the safety of employees, on raw material supply and cash management. Within a few days, a hand disinfectant gel was brought to market maturity and nationwide availability of up to 6 million units was ensured via pharmacies by the beginning of June. Administrative districts and medical staff were also supplied with it.
Parallel development of further Corona prophylaxis products had top priority. Two milestones followed at the end of 2020: the scientific confirmation of a limited virucidal hand cream and clinical results for prophylaxis by means of a medical mouth and throat rinse. Since March 2020, Dr. Wolff research has been examining laboratory and clinical data on physical Corona protection provided by this specially developed mouth and throat rinse. In two laboratory studies and a first clinical application study on 34 COVID-19 patients, a possible prophylaxis could be achieved by significantly reducing the viral load by up to 90 percent. The results were submitted as a paper.
Another placebo-controlled clinical trial launched in February investigated the cumulative effect of several days’ use in COVID-19 patients. As was announced yesterday, an international group of authors has now come to the conclusion in their study[1] that the oral cavity is the decisive gateway for the virus to enter the body. The virus enters blood vessels via the saliva and thus reaches the lungs. Researchers concluded: “Reducing the viral load in mouth and throat can prevent severe disease progression.” Using a mouth rinse is thus the recommended corona prophylaxis.
“We are driven by the goal to complement existing Corona prophylaxis options in the best possible way and to make an effective contribution to reducing the viral load. This is not to replace vaccination, but the so-far existing steps and measures do not suffice. We need further effective means to contain the virus, to better protect ourselves and others and to break transmission chains,” says Eduard R. Dörrenberg, managing partner of Dr. Wolff Group, describing Germany’s slow progress in fighting the pandemic compared to other countries and adds: “The measures taken so far are the result of a singular approach: First, distance and hygiene rules were introduced. Then, after initial scepticism, masks had to be worn as demanded by hygienists (social distancing, hygiene and room ventilation rules), before we started striving for vaccinations and now also for testing strategies.”
Paul Ehrlich Institute also confirms, according to their president Prof. Klaus Cichutek: “If the virus load in aerosols is significantly reduced because of a reduced viral load in the throat, this might lead to less or no contact persons at all being infected.” [2]
New potentials for Dr. Wolff in drug development, familiar brands, modern products and further digitalisation
In the field of in-house drug research, Dr. Wolff is expected to soon offer a solution to pathological sweating in the armpits (so-called primary axillary hyperhidrosis): after several years of development, the pharmaceutical division submitted the application for approval of the drug, which is unique in the EU, in the decentralised procedure (DCP) for Germany, Sweden and a further 9 EU countries at the end of February. Dr. August Wolff will market the cream directly or in cooperation with experienced partners in these countries. The application for approval of the formulation containing 1% Glycopyrronium Bromide (GPB) against pathological sweating in the armpits is based on data from several clinical studies, with the phase 3a part already published in the renowned British Journal of Dermatology.[3]
Outlook
“Corona is still here!” This is the conclusion after more than a year of living with the pandemic. Asia could emerge as the winner, as they have succeeded in controlling the pandemic and in developing strategies to normalise public life and the handling of the pandemic. “We have already seen a noticeable normalisation of our Asian business with the first relaxations, for example in China,” confirms Dörrenberg, who paid a visit to the Dr. Wolff branch in Singapore at the beginning of 2021 after a year’s abstinence. “The differences are very drastic. Enormous transparency and contact tracing by means of consistent check-in processes at buildings, for example, temperature measurements and Corona tests, have brought normalisation and control to pandemic development in Singapore for months already. In Europe, on the other hand, this will take longer, and we may even lose ground. I therefore expect a slower development of our business in the second quarter,” Dörrenberg predicts.
The value and benefit of strong brands, efficient products, increasing digitalisation and own data will rise all the more. Reaching consumers – who act in an increasingly individualised manner – with the right product at the right time will make the difference in medicine and cosmetics. It will be about shaping future ways of communication and work. According to Dörrenberg the economy has already made enormous progress as to that, but there is still room for improvement: “Politics must finally follow suit, I would appreciate more transparency, more competence and more digitalisation. We must no longer just wait and see. First, we did not protect the elderly sufficiently. Now we are depriving our children of education because we fail at organising, digitalising and structuring education in an up-to-date manner. Just recently, the Scientific Advisory Council of the German Ministry of Economics described the conditions in public authorities as ‘archaic’.” Nevertheless, Dr. Wolff is optimistic about the future. We hope and wish for improved political framework conditions, with the focus remaining on research, innovations and digitalisation.
About our brands
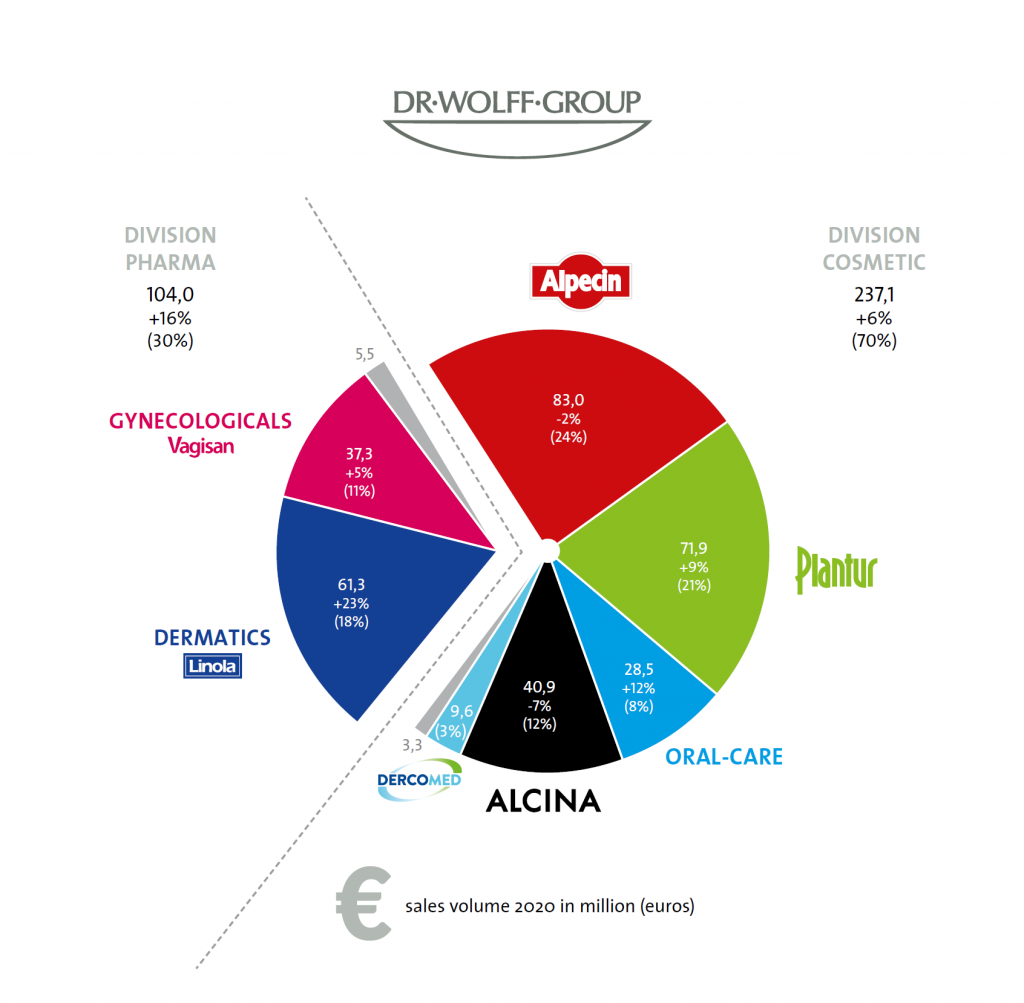
Pharmaceutical division:
Linola:
In addition to increased demand for hand cream due to skin damage caused by washing hands and using disinfectant solutions more often, Linola is growing overall with medical skin care. The introduction of the hand hygiene series under the roof of Linola Sept brand, including hand cleanser, hand disinfectant gel and an anti-Corona hand cream strengthens the positive development. An additional component of effective Corona prophylaxis is the mouth and throat rinse, which has been available in pharmacies since January.
Vagisan:
Currently, about eight million women in Germany are going through menopause – as many as never before. Dr. Wolff’s gynaecological products have been contributing considerably for years, for example in hormone-free therapy. In the past year, the brand continued to grow both at home and abroad. Nationally, it is the sexologist Ann-Marlene Henning who, as brand ambassador, openly addresses difficult topics such as vaginal dryness and advises affected women.
Cosmetic division:
Oral care:
Research with far-reaching studies on modern caries prophylaxis and education around European regulation on the dosage of fluoridated toothpaste are a main focus in the oral care sector. Within the past 10 years, more in-depth research has been conducted worldwide on hydroxyapatite and its positive influence on dental health than on any other oral care ingredient. Numerous studies and publications show that hydroxyapatite is a real alternative to fluoride. In particular, the fluoride-free children’s tooth paste Kinder Karex increased in sales and is being consciously used by parents as an alternative to fluoride-containing dental care. With a 12 per cent increase, the next step is internationalisation.
Plantur:
The brand against female hair loss and for hair growth is up nine percent. During lockdown, products with a colouring effect against visible hairlines hit a nerve. Especially an individual, content-coordinated digital communication determined by relevant topics reached the target group. Young women with long hair are addressed with the new Plantur 21 series. A research group led by Dr. Gil Westgate (University of Bradford) also confirmed that caffeine was the best-researched active plant-based ingredient against hair loss.
Alpecin:
About every second man is affected by hair loss in the course of his life. In 80 percent of cases, the cause is hereditary. Alpecin caffeine shampoo is made for slowing down this process and providing effective prevention. Educating young men is therefore crucial. Whether this is due to a lack of sporting opportunities or more home office time: the German shampoo market declined by five percent in 2020. Alpecin performed better than the market, lost two percent compared to the previous year and remained stable in times of crisis.
Alcina:
Hairdressing business has undergone two lockdowns so far. In 2020, Alcina focused specifically on products that have been in demand of consumers in the current situation or that simplify work in the salons, e.g., with regard to hygiene and service. By means of videos and numerous digital offers, the brand took the opportunity to support its B2B customers in the best possible way in areas such as salon management or craft training. New digital formats were also created for end consumers to promote customer loyalty. Colouring and care products contributed more to sales of almost 41 million euros in the financial year than, for example, decorative cosmetics.
[1] Lloyd-Jones et al. The COVID-19 Pathway: A Proposed Oral-
Vascular-Pulmonary Route of SARS-CoV-2 Infection and the Importance of Oral Healthcare Measures. Journal of Oral Medicine and Dental Research. 2021. Genesis-JOMDR-2(1)-S1 Volume 2 | Issue 1 Open Access
[2] ARD-Morgenmagazin April 14, 2021
[3] Abels C, Soeberdt M, Kilic A, Reich H, Knie U, Jourdan C, Schramm K, Heimstaedt-Muskett S, Masur C, Szeimies RM. A 1% glycopyrronium bromide cream for the topical treatment of primary axillary hyperhidrosis: Efficacy and Safety Results from a Phase 3a Randomised Controlled Study. Br J Dermatol. 2021 Jan 14. doi: 10.1111/bjd.19810